FDA 21 CFR Part 11 compliance -- electronic records and signatures done right
Map every section of 21 CFR Part 11 to your electronic record-keeping system. Validated platform, immutable audit trails, compliant electronic signatures, and granular access controls -- all in one platform.
No credit card required · No long-term contract · UK-based support · Live in 4 weeks
What is 21 CFR Part 11?
21 CFR Part 11 is a United States FDA regulation that establishes the criteria under which electronic records and electronic signatures are considered trustworthy, reliable, and equivalent to paper records and handwritten signatures. It applies to any FDA-regulated industry using electronic systems to create, modify, maintain, archive, retrieve, or transmit records.
The regulation is divided into three subparts: Subpart A (General Provisions), Subpart B (Electronic Records), and Subpart C (Electronic Signatures). Key requirements include system validation, complete audit trails, electronic signatures with intent, authority checks, and device checks. Compliance is mandatory for pharmaceutical, biotech, medical device, and food manufacturers regulated by the FDA.
Key Facts
- Enacted
- 1997 (21 CFR Part 11)
- Enforced by
- US Food & Drug Administration (FDA)
- Applies to
- Pharma, biotech, medical devices, food, cosmetics
- Key requirements
- System validation, audit trails, e-signatures, access controls, record integrity
- EU equivalent
- EU GMP Annex 11 (Computerised Systems)
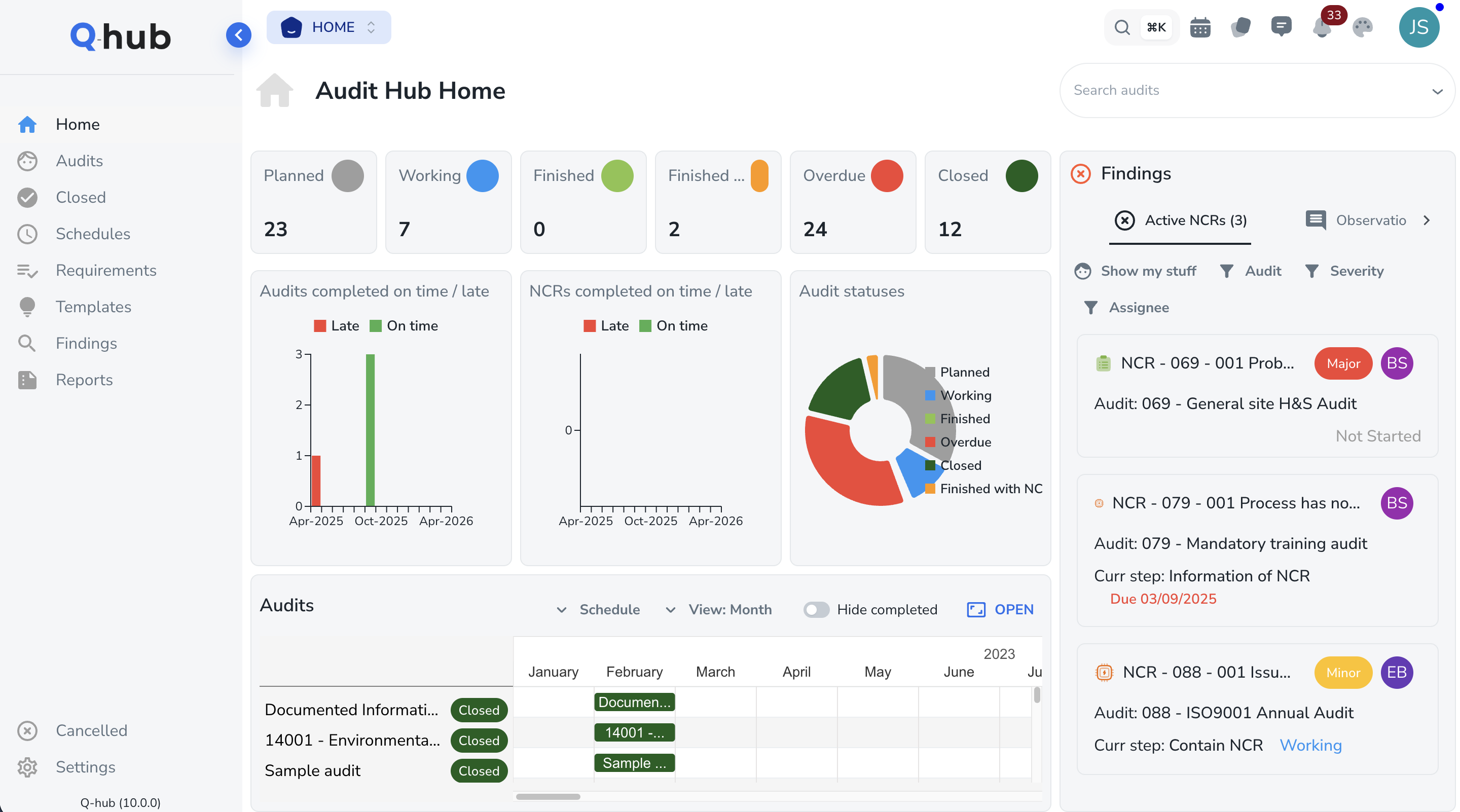
How Q-Hub maps to 21 CFR Part 11 requirements
Subpart B -- Electronic Records
Subpart C -- Electronic Signatures
Be inspection-ready for FDA audits and EU GMP Annex 11
FDA inspection readiness
Every electronic record is backed by an immutable audit trail. When FDA investigators request evidence, pull complete record histories -- who did what, when, and why -- in seconds, not days.
EU GMP Annex 11 alignment
Q-Hub is designed to satisfy both FDA 21 CFR Part 11 and EU GMP Annex 11 requirements simultaneously. One validated system, two regulatory frameworks covered -- ideal for companies selling into both US and EU markets.
Validation documentation included
Q-Hub provides IQ/OQ/PQ documentation, validation protocols, traceability matrices, and risk assessments as part of your implementation. Reduce validation effort by 50% compared to building documentation from scratch.
SOPs, batch records, and change control -- fully electronic
21 CFR Part 11 requires that electronic records are created, modified, maintained, and archived under controlled conditions. Q-Hub handles the full document lifecycle: version-controlled SOPs, electronic batch records, deviation reports, and change control workflows -- all with complete audit trail and compliant electronic signatures.
- Version-controlled SOPs with full audit trail
- Electronic batch records replacing paper
- Change control workflows with impact assessment
- Deviation tracking with CAPA linkage
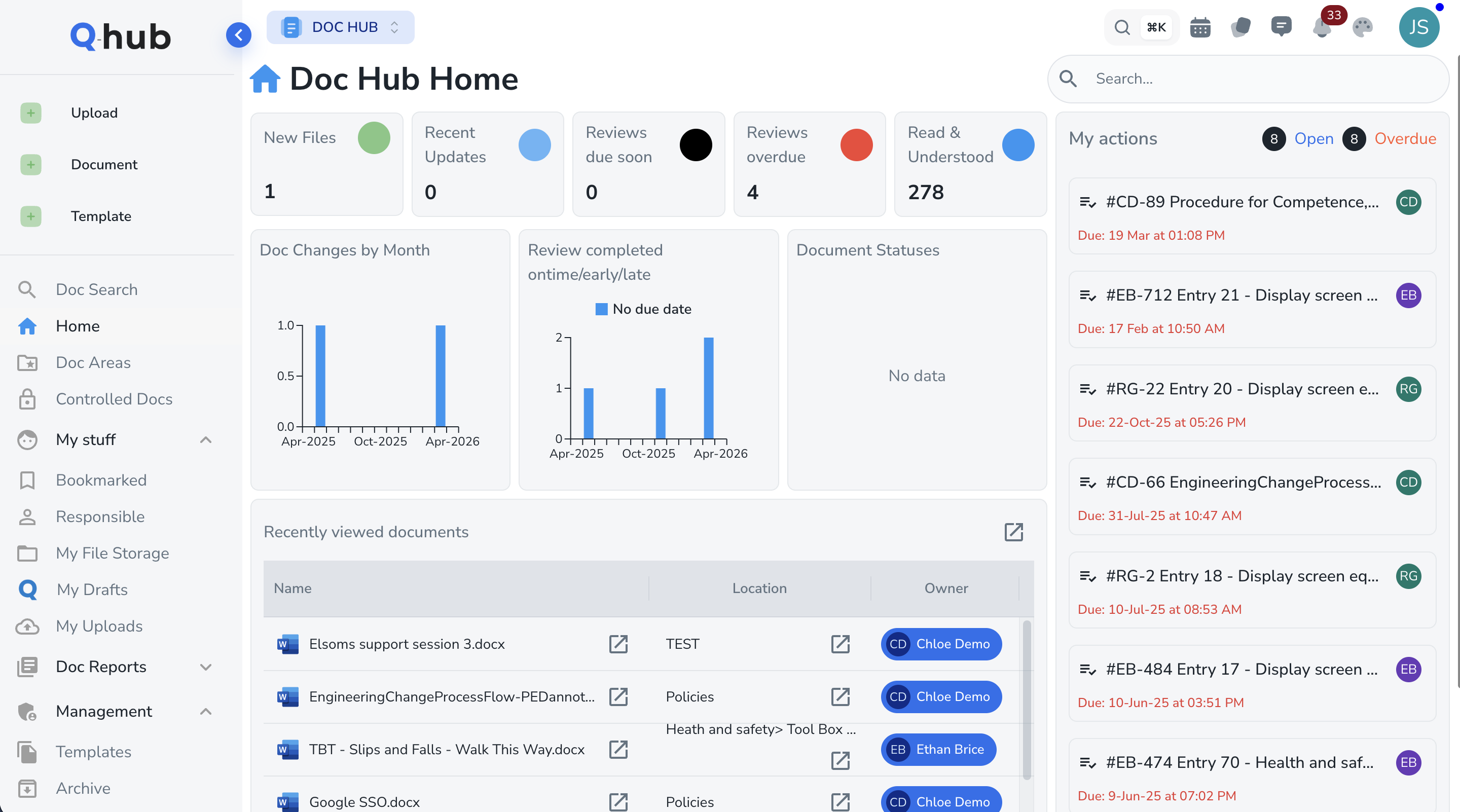
Electronic signatures that meet FDA requirements
Part 11 demands that electronic signatures include the printed name of the signer, the date and time of signing, and the meaning of the signature (e.g. review, approval, responsibility). Q-Hub captures all three elements and cryptographically binds each signature to its associated record, making it tamper-evident and legally defensible.
- Name, date, time, and meaning captured on every signature
- Signatures cryptographically bound to records
- Individual user accounts -- no shared logins permitted
- Password complexity, expiry, and account lockout controls
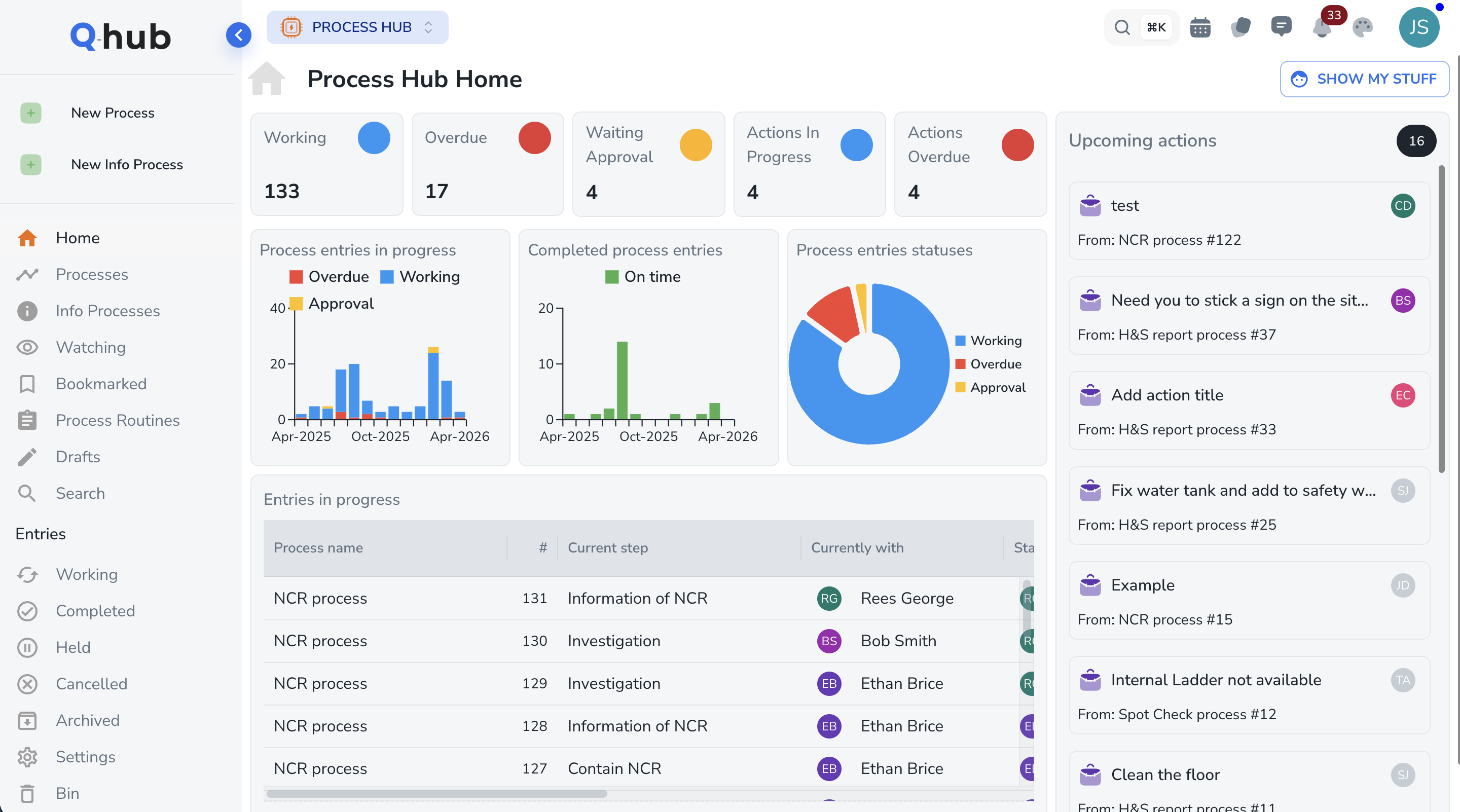
Results from Q-Hub customers achieving 21 CFR Part 11 compliance
21 CFR Part 11 compliance timeline with Q-Hub
Gap analysis & risk assessment
We assess your current systems against Part 11 requirements and identify gaps. Risk-based approach determines validation scope.
System configuration & IQ/OQ
Configure Q-Hub for your workflows. Execute Installation Qualification and Operational Qualification protocols.
PQ & user training
Performance Qualification with your live data. Train all users on compliant electronic record-keeping and e-signature procedures.
Go-live & validation report
Full production deployment with validation summary report. Ongoing periodic review and revalidation support included.
FDA 21 CFR Part 11 FAQs
What is 21 CFR Part 11?
21 CFR Part 11 is a regulation issued by the US Food and Drug Administration (FDA) that defines the criteria under which electronic records and electronic signatures are considered trustworthy, reliable, and equivalent to paper records and handwritten signatures. It was enacted in 1997 and applies to all FDA-regulated industries including pharmaceuticals, biotechnology, medical devices, and food manufacturing. The regulation covers system validation, audit trails, access controls, and electronic signature requirements.
Does Q-Hub comply with Part 11?
Yes. Q-Hub is designed from the ground up to support 21 CFR Part 11 compliance. The platform provides all the technical controls required by the regulation: system validation with IQ/OQ/PQ documentation, immutable audit trails on every record, compliant electronic signatures with name, date, time, and meaning, role-based access controls with MFA, and cryptographic binding of signatures to records. Q-Hub also provides the validation documentation you need to demonstrate compliance to FDA inspectors.
What about EU GMP Annex 11?
EU GMP Annex 11 is the European equivalent to 21 CFR Part 11, governing computerised systems used in GMP-regulated environments. While the two regulations have different structures, they share similar requirements around system validation, data integrity, audit trails, and access controls. Q-Hub satisfies both frameworks simultaneously, making it ideal for organisations that manufacture or distribute products in both the US and EU markets. One validated system covers both regulatory frameworks.
How does Q-Hub handle electronic signatures?
Q-Hub electronic signatures comply with Subpart C of 21 CFR Part 11. Every signature captures the signer's full name, the date and time of signing, and the meaning of the signature (such as "authored", "reviewed", or "approved"). Signatures are cryptographically bound to the record they sign, ensuring they cannot be transferred, copied, or falsified. Each user has a unique account with no shared logins, and authentication uses username plus password with configurable password complexity, expiry, and account lockout policies.
What validation documentation does Q-Hub provide?
Q-Hub provides comprehensive validation documentation as part of your implementation, including: Validation Plan, User Requirements Specification (URS), Functional Requirements Specification (FRS), Installation Qualification (IQ) protocols, Operational Qualification (OQ) protocols, Performance Qualification (PQ) protocols, traceability matrices linking requirements to test cases, risk assessments, and a Validation Summary Report. This documentation reduces validation effort by approximately 50% compared to building everything from scratch.
Can Q-Hub replace paper batch records?
Yes. Q-Hub enables you to move from paper-based batch records to fully electronic batch records that comply with 21 CFR Part 11. Electronic batch records in Q-Hub include all the controls required by the regulation: audit trails recording every creation and modification, electronic signatures for review and approval steps, access controls ensuring only authorised personnel can create or modify records, and the ability to generate legible, accurate copies in both human-readable and electronic form. Many of our pharmaceutical customers have eliminated paper batch records entirely.
Ready to achieve 21 CFR Part 11 compliance?
See how Q-Hub maps to every section of Part 11 and gets you inspection-ready in 12-16 weeks.
