Pharmaceutical QMS Software
Pharmaceutical companies trust Q-Hub to replace paper-heavy change control, manual validation overhead, and fragmented audit trails with one validated platform. GxP and FDA 21 CFR Part 11 compliant. Electronic signatures, complete traceability, and inspection-ready documentation. Live in 4 weeks.
No credit card required · No long-term contract · UK-based support · Live in 4 weeks
Trusted by pharmaceutical manufacturers, CMOs, and biotech companies
Sound familiar?
Validation consumes months of effort
Every new system requires IQ/OQ/PQ protocols, traceability matrices, and risk assessments. By the time validation is complete, the business has already moved on to the next compliance fire.
Change control drowns in paper
Change requests are routed via email, impact assessments live in Word documents, and approval signatures are collected on wet-ink forms. A single change can take weeks to close when it should take days.
Audit trail gaps risk regulatory findings
When the MHRA or FDA inspector asks who accessed a document, when it was modified, and why — the answer should be instant. Instead, your QA team spends hours piecing together evidence from multiple systems.
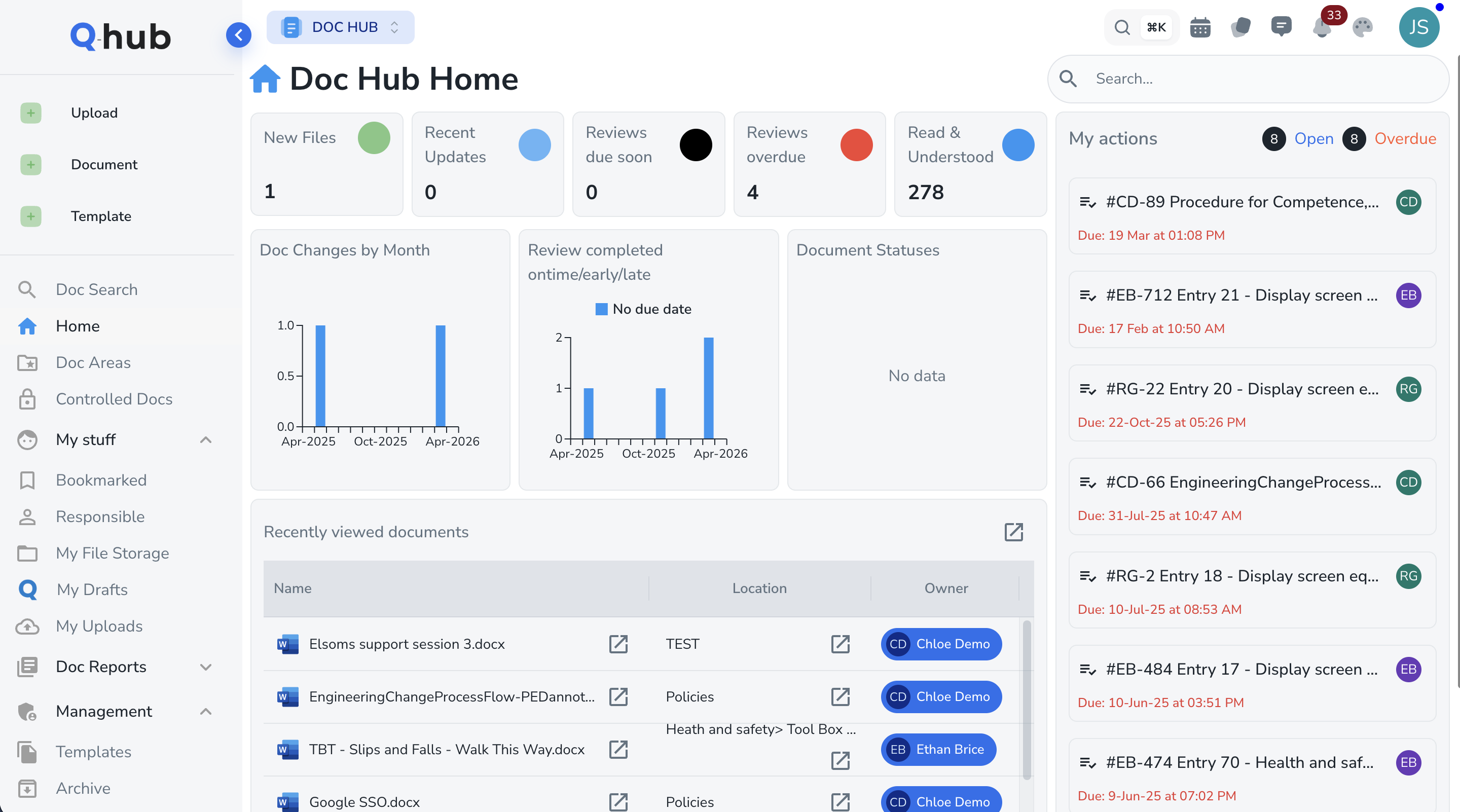
One validated platform for pharmaceutical quality management
Validated Document Control
SOPs, batch records, and quality manuals — version controlled with multi-level approval workflows and electronic signatures. Every document action is captured in an immutable audit trail. FDA 21 CFR Part 11 and EU GMP Annex 11 compliant out of the box.
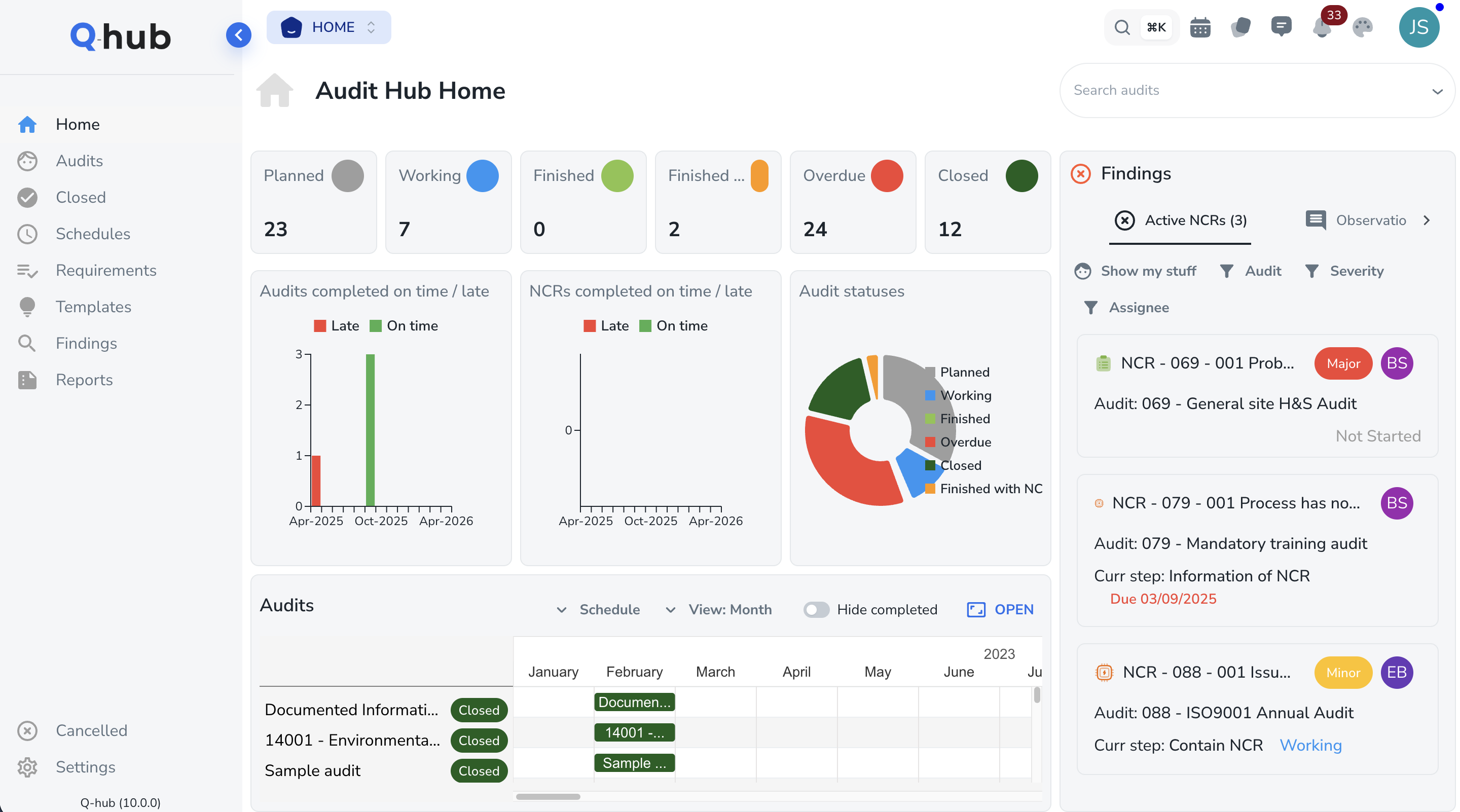
GxP Audits & Inspection Readiness
Schedule and conduct internal GMP audits, supplier qualification audits, and regulatory inspection preparation reviews. Findings generate CAPA automatically. Complete audit history with evidence attachments provides inspection-ready documentation at the click of a button.
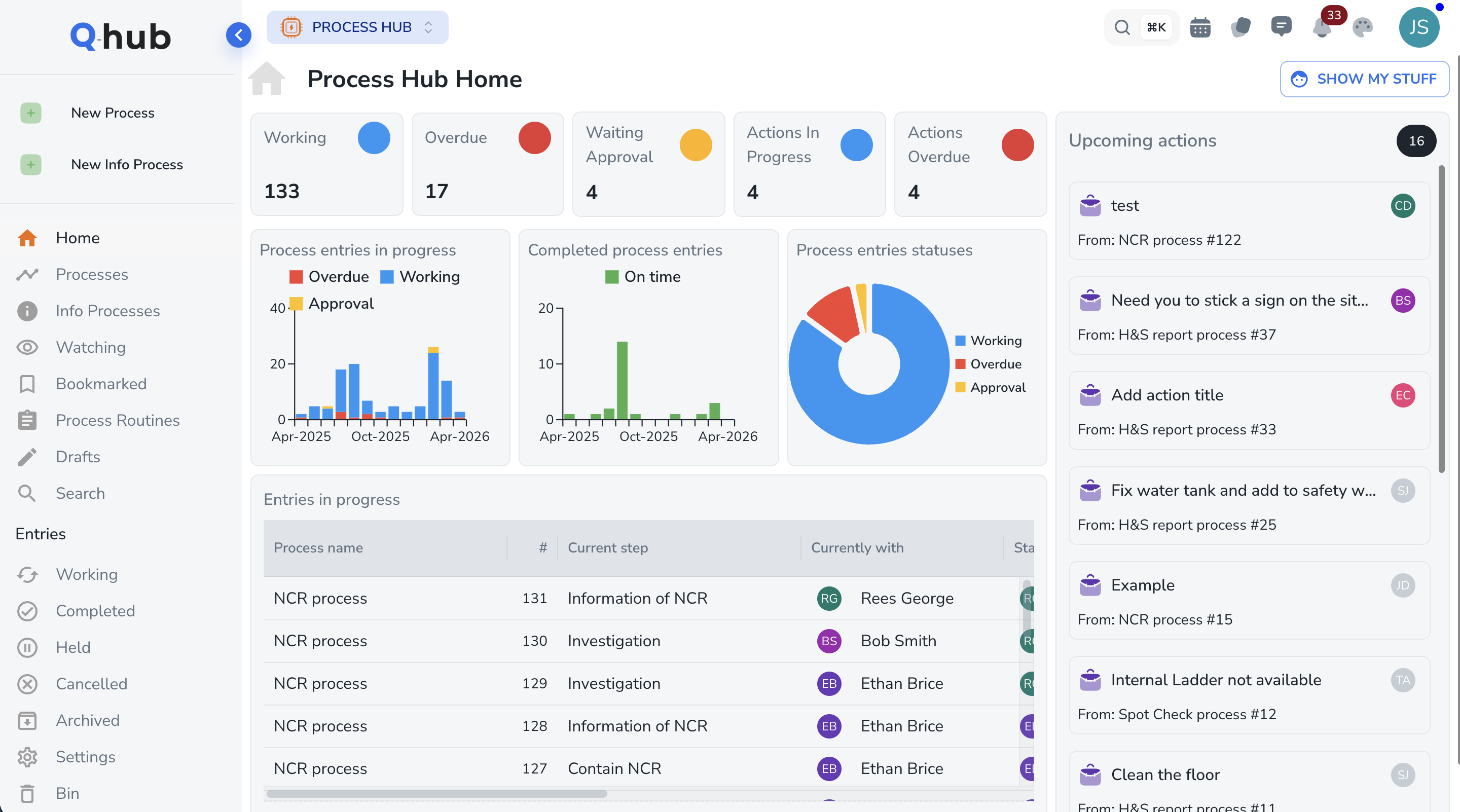
Change Control & Deviation Management
Manage the full change control lifecycle — request, impact assessment, approval, implementation, and effectiveness review. Deviations are logged with root cause analysis and linked CAPA. Every step is electronically signed and audit-trailed for regulatory confidence.
Built for pharmaceutical regulations
The numbers from our pharma customers
Based on average results from Q-Hub pharma customers. Individual results may vary. Read customer stories