CAPA Management Software -- Close the Loop on Non-Conformances
Ensure every non-conformance is investigated, corrected, and verified -- with full audit trail. From 8D investigations to effectiveness verification, Q-Hub keeps your CAPA process on track.
No credit card required · No long-term contract · UK-based support · Live in 4 weeks
Still tracking corrective actions in spreadsheets and email chains?
See how Q-Hub CAPA Management transforms your non-conformance process.
Without Q-Hub
With Q-Hub CAPA Management
14-day free trial · No credit card · Live in 4 weeks
See CAPA Management in action
Watch a 2-minute walkthrough of non-conformance logging, root cause analysis, and effectiveness verification.
Everything you need to close the loop on every non-conformance
Six core capabilities that replace spreadsheet CAPA logs, email follow-ups, and manual tracking.
8D Methodology
Structured eight-discipline workflow guiding teams from problem definition through to verified close-out.
Root Cause Analysis
Built-in 5-Why, fishbone (Ishikawa), and fault tree analysis tools to identify true root causes.
Action Tracking
Assign corrective and preventive actions with owners, deadlines, evidence requirements, and escalation rules.
Effectiveness Verification
Schedule independent verification reviews to confirm corrective actions resolved the root cause.
Trend Analysis
Dashboards highlighting recurring non-conformance categories, root cause types, and departmental patterns.
Cross-Module Integration
CAPAs auto-triggered from audits, incidents, complaints, and management reviews with full traceability.
Take the product tour
Explore CAPA Management end-to-end, from non-conformance capture to verified close-out.

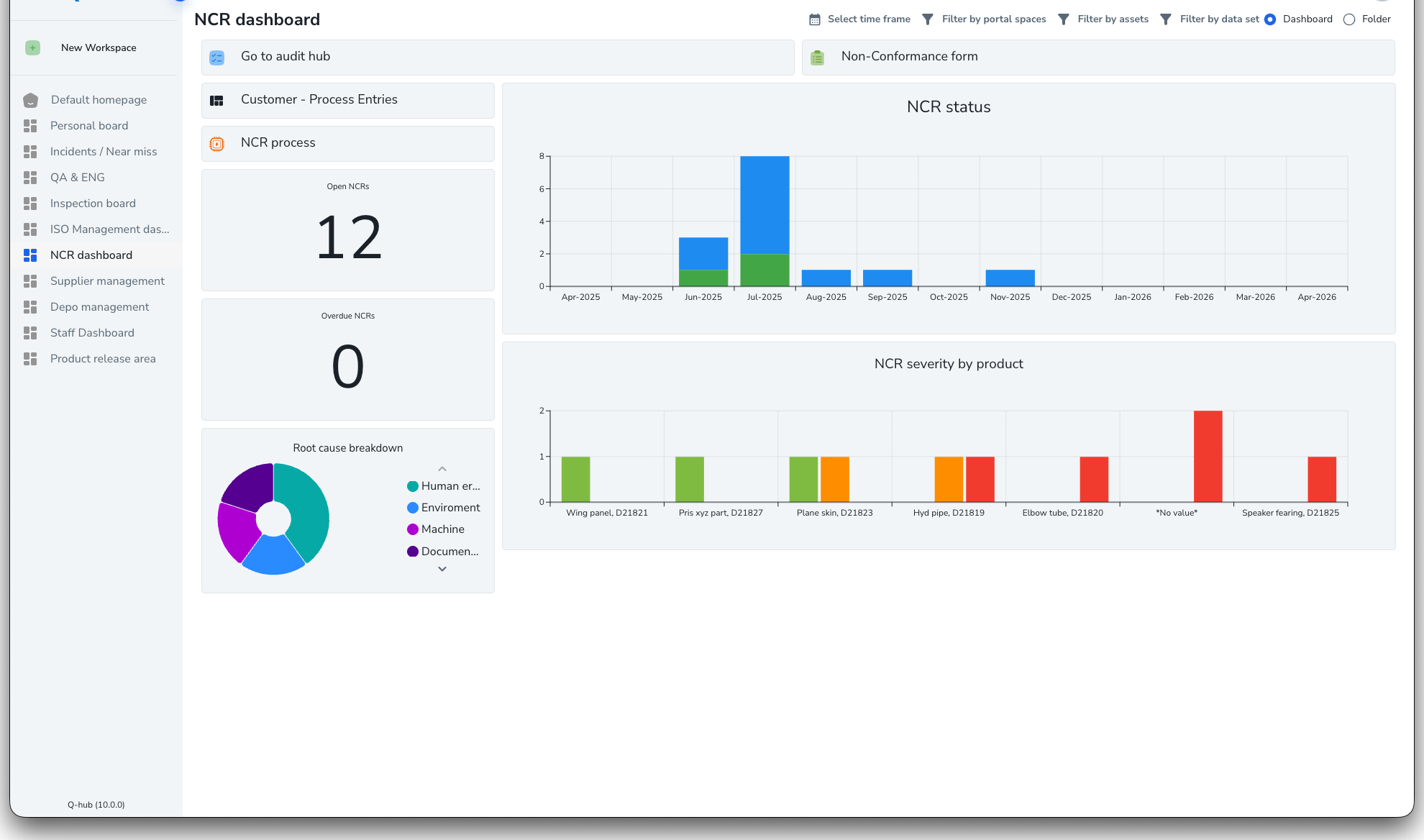
Log and categorise every non-conformance
Capture non-conformances from any source -- audits, incidents, customer complaints, or management reviews. Each NC is categorised by type, severity, and department, creating a searchable register with full audit trail.
- Auto-populated from audits, incidents, and complaints
- Categorise by type: process, product, supplier, system
- Severity grading: critical, major, minor, observation
- Searchable register with filters and export
Identify true root causes with structured tools
Use built-in 5-Why analysis, fishbone diagrams, or fault tree analysis to dig beyond symptoms. Each RCA is documented with evidence and linked to the corrective action plan.
- Interactive 5-Why drill-down with evidence fields
- Fishbone (Ishikawa) diagram builder
- Fault tree analysis for complex failures
- RCA linked directly to corrective actions
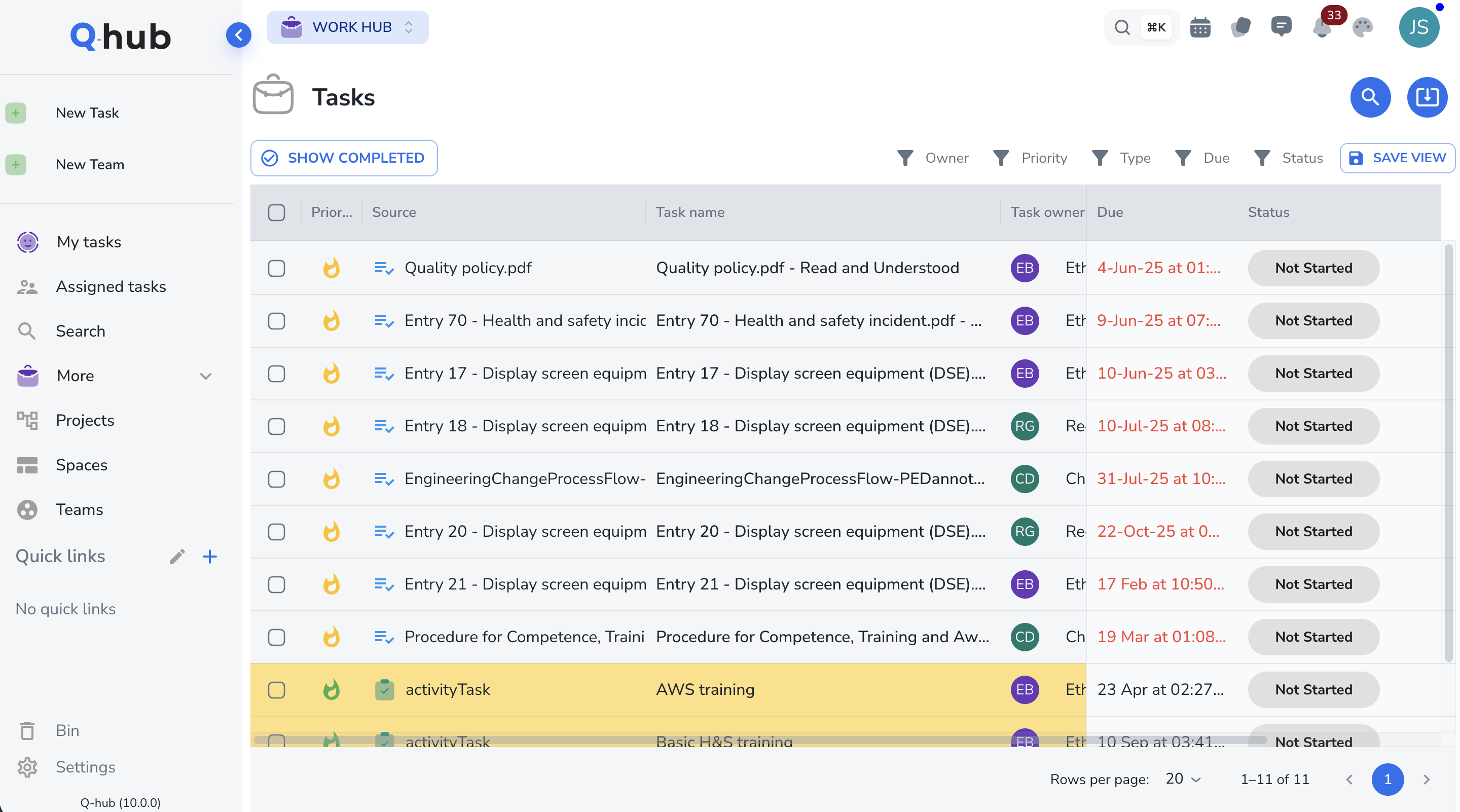
Plan and assign corrective and preventive actions
Create action plans with assigned owners, target dates, and required evidence. Both corrective (fix the problem) and preventive (stop it recurring) actions are tracked separately with independent deadlines.
- Separate corrective and preventive action streams
- Owner assignment with email notifications
- Evidence upload requirements per action
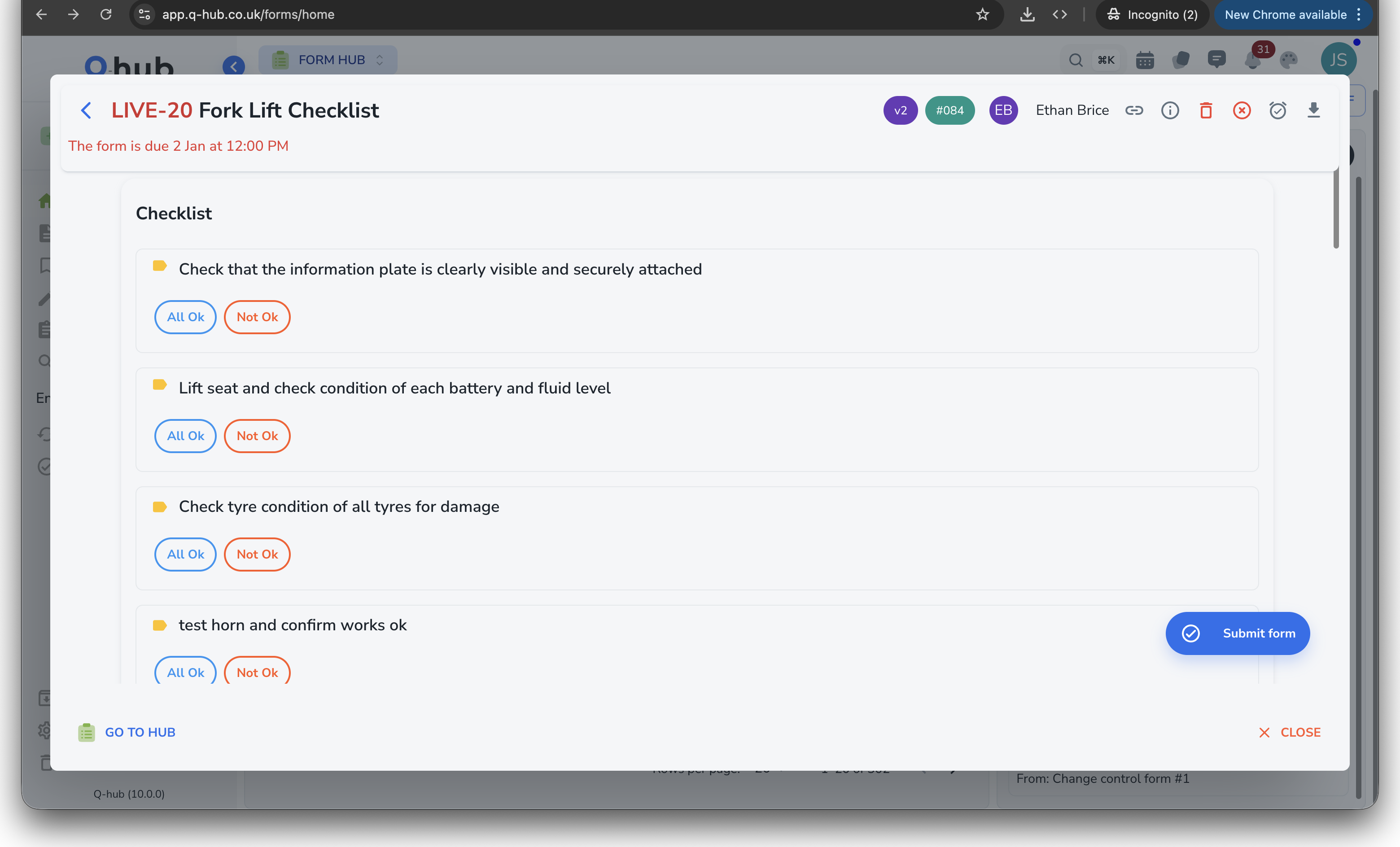
Verify that actions actually resolved the issue
After corrective actions are implemented, an independent reviewer confirms effectiveness. CAPAs cannot be closed without documented verification evidence -- meeting auditor expectations every time.
- Scheduled verification step with independent reviewer
- Mandatory evidence before closure is permitted
- Re-open workflow if verification fails
Spot trends and prevent recurring non-conformances
Dashboards show CAPA volumes by category, root cause type, department, and time period. Identify systemic issues before they become major findings at your next external audit.
- NC trends by category, department, and source
- Root cause Pareto analysis
- Closure rate tracking and overdue alerts
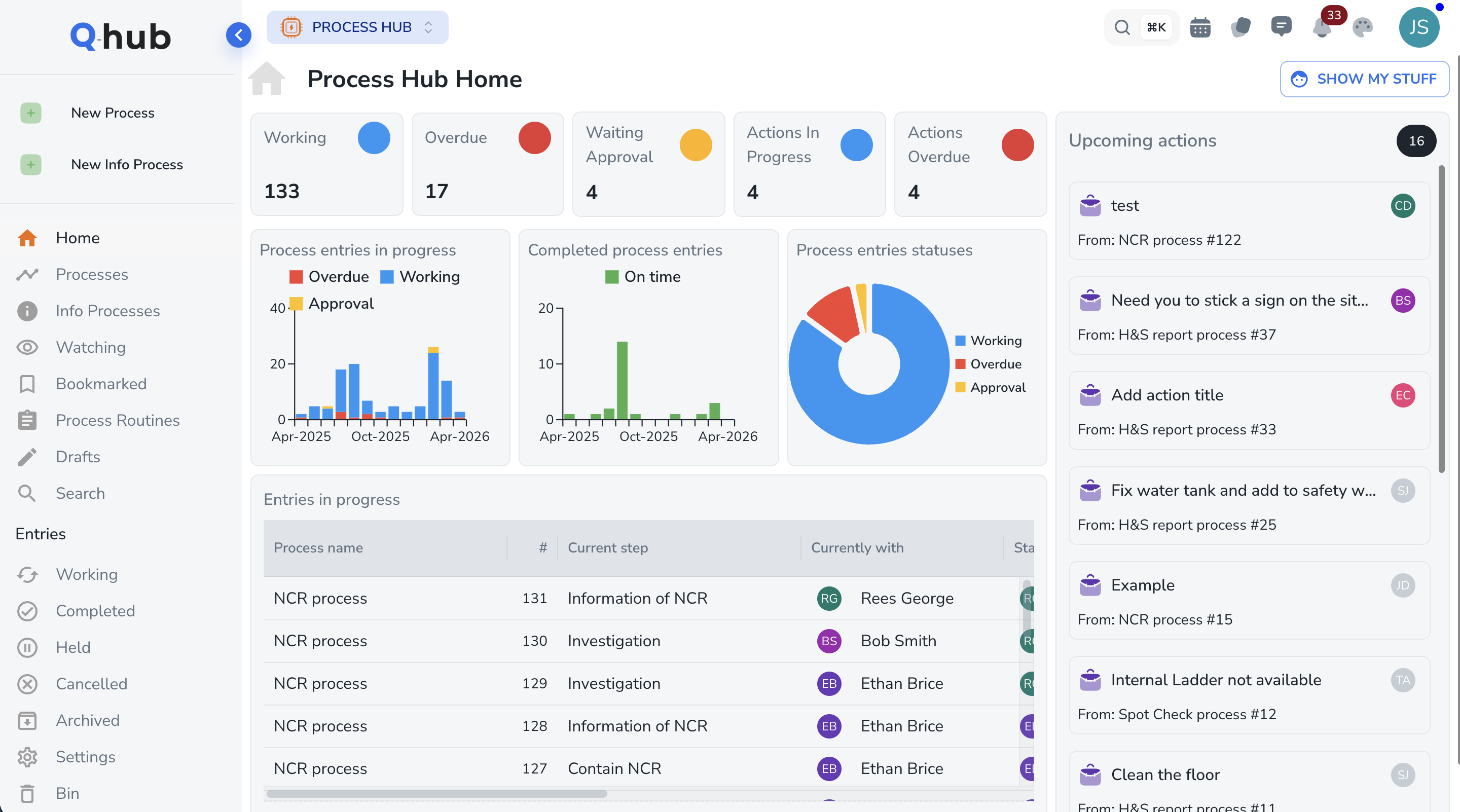
From non-conformance to verified closure in five steps
A structured CAPA lifecycle that meets ISO requirements and auditor expectations.

Investigate root causes -- not just symptoms
Quality managers and investigators use built-in 5-Why and fishbone tools to systematically drill down to the true root cause of every non-conformance. Each investigation is documented with evidence, linked to the corrective action plan, and forms part of the permanent quality record.
- 5-Why drill-down with branching paths
- Fishbone diagram builder with standard categories
- Attach evidence: photos, documents, test results

Track every action to verified closure
Corrective and preventive actions are tracked in a single dashboard with clear ownership, deadlines, and progress indicators. Automatic escalation rules ensure overdue actions are flagged to management before they become audit findings. Nothing falls through the cracks.
- Assigned owners with deadline reminders
- Automatic escalation for overdue actions
- Evidence requirements before action can be completed
Calculate your CAPA time savings
See how many hours CAPA Management could save your quality team every month.
Connects with the tools you already use
CAPA Management integrates with your existing ecosystem via REST API, webhooks, and pre-built connectors.
CAPA Management vs the alternatives
See why quality teams switch from spreadsheets and email-based CAPA tracking to Q-Hub.
| Capability | Q-Hub CAPA Management | Spreadsheet Tracking | Email-Based Process |
|---|---|---|---|
| Structured 8D workflow | |||
| Built-in root cause analysis tools | |||
| Auto-triggered from audits/incidents | Manual | ||
| Effectiveness verification workflow | Ad hoc | ||
| Deadline tracking & escalation | Manual | ||
| Trend analysis & Pareto charts | Limited | ||
| Full audit trail |
Trusted by quality teams worldwide
Frequently asked questions
Works even better together
CAPA Management is part of the Q-Hub integrated platform. Combine it with these modules for end-to-end compliance.
See CAPA Management in action
Book a personalised demo tailored to your CAPA process. See exactly how your non-conformances would flow through Q-Hub.
No credit card required · No long-term contract · UK-based support · Live in 4 weeks
